Based on the long-term tracking and research of Ruiou Technology, the main reasons for the unapproved active substances in the EU are as follows:
❶ Active substances are too harmful
Based on Regulation (EU) 1107/2009, if a substance is classified as a CMR substance (a teratogenic, carcinogenic, mutagenic substance classified as 1A or 1B), or as an endocrine disruptor, in terms of human health, such Substances can be banned without risk assessment. Although there is still a gap in the regulations, that is, in some cases, if the use of the product will not cause human exposure, it is still possible to approve it, but for field pesticides used outdoors, this is basically impossible.
In terms of the environment, the standard for banned substances is whether a substance will be identified as POP, PBT or vPvB. In addition to toxicity, additional considerations on the environmental side are the persistence and bioaccumulation of the substance in the environment.
❷ The risk of using pesticide products is uncontrollable
A risk assessment cannot show that a preparation containing an active substance can be used safely under reasonable conditions.
For example, neonicotinoid pesticides that are banned due to uncontrollable product use risks.
In recent years, in order to protect bees, the EU has banned most of the neonicotinoids, such as thiamethoxam, clothianidin, imidacloprid, thiacloprid, etc.
By looking at the CLP classification of these substances, we can find that in addition to thiacloprid, several other substances do not meet the banned substances in the CLP classification. Only because the risk assessment showed that the use of these substances was uncontrollable for the risks to bees, so officials made the decision to ban the substances.
❸ Incomplete application dossier
The applicant did not submit sufficient data, and the official did not approve the evaluation due to lack of data.
❹ No support for active substance re-evaluation
Including: no one supports the re-evaluation of a substance at the beginning and the supporter of the re-evaluation withdraws the application in the middle of the review.
For example, the ban of glufosinate-ammonium caused a sensation in the early years.
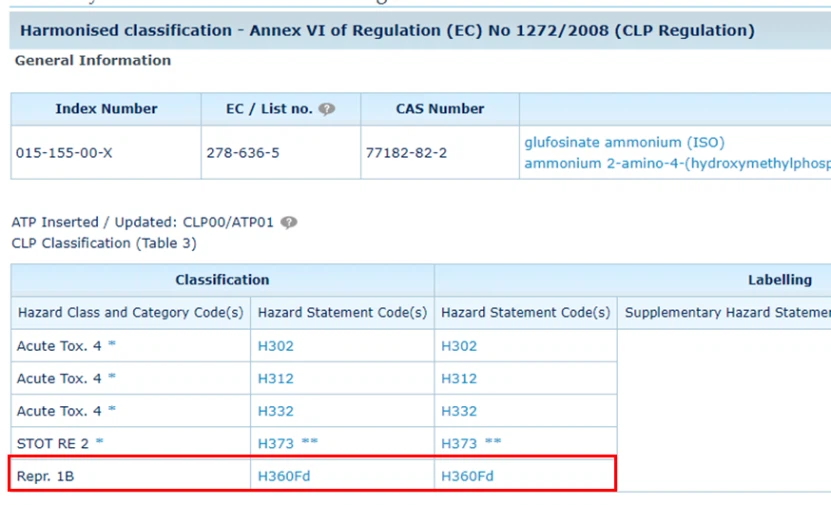
Based on the information published by the EU, the re-evaluation application and complete dossier for glufosinate-ammonium were submitted in 2014 and 2016, respectively, but during the dossier review process, the applicant did not choose to continue the defense, which ultimately resulted in the substance not being approved.
� With such a popular product, the market prospect is very good. Why did the applicant choose to give up the defense halfway after submitting the case?
The most likely reason is because of the CLP classification of glufosinate. In terms of reproductive toxicity, glufosinate-ammonium is classified as 1B. According to Regulation 1107, for this classification, unless the applicant can ensure that under the recommended conditions of use, the use of the product will cause little human exposure, otherwise the final result is still will be disabled.
For field pesticides, human exposure is inevitable, so glufosinate-ammonium does not meet the approval conditions for EU pesticide active substances in essence, and the applicant should also stop losses in the middle of the re-evaluation because of this, and choose not to continue to support. The ban caused by material hazards and product use risks is more difficult to overturn, but the latter two reasons are more of the ban caused by the applicant giving up support due to his own multi-factor considerations.










